Researchers from the laboratory of an Iconeus co-founders have used functional ultrasound imaging (fUSI) in awake mice to show a correlation between opioid-induced changes in brain functional connectivity and analgesia. The high-sensitivity images were acquired without sedation and craniotomy, positioning fUSI as a potentially valuable tool in the development of safer and more effective pain therapies.
The ongoing opioid crisis drives the development of potent analgesics with reduced side-effects. However, this effort is hampered by gaps in our understanding of how existing drugs modulate brain function at the systems level.
A promising approach lies in advanced brain imaging techniques, which map drug-induced changes in neural activation and connectivity patterns. By identifying these signatures, researchers can generate testable hypotheses, and uncover the underlying mechanisms of action.
Now, in a paper published in PNAS, a team of French researchers from Inserm – including co-founders of Iconeus – have pioneered a robust ‘pharmaco-fUS’ approach in mice. Using functional ultrasound imaging (fUSI), they successfully identified opioid-induced alterations in functional connectivity offering fresh insights into the neurobiological basis of opioid analgesia.
Opioid-induced changes in Functional Brain Connectivity
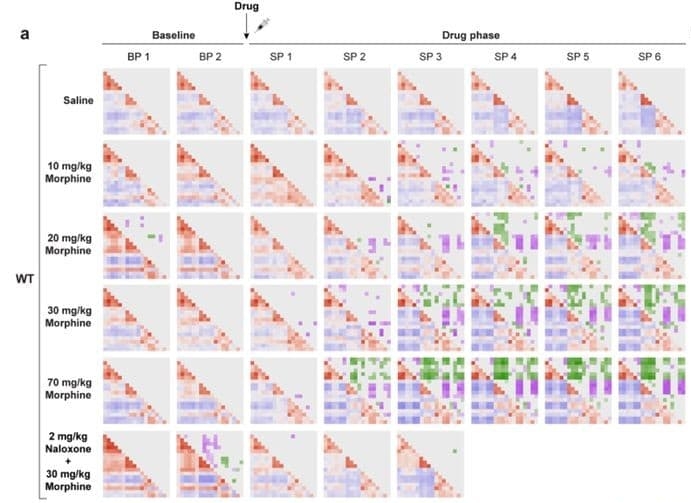
A research team, led by PhD student Jean-Charles Mariani and guest scientist Dr Andrea Kliewer in the group of Dr Zsolt Lenkei at the Institute of Psychiatry and Neurosciences of Paris (Université Paris Cité), used a linear probe to capture coronal-plane transcranial images of awake mice following treatment with morphine, fentanyl, methadone or buprenorphine.
They found that these drugs induced a set of significant but gradually-evolving changes in functional connectivity between cortical and subcortical brain regions. These changes were specific to the activation of the µ-opioid receptor, were dose-dependent, and diminished as tolerance developed with prolonged drug exposure.
Further experiments showed that the reorganization of functional connectivity, along with the whole-brain activation of the µ-opioid receptor, occurred on a similar timescale than the development of analgesia. However, this process evolved much more slowly than the rapid changes in locomotion, respiration rate and regional cerebral blood volume (CBV) changes induced by morphine – suggesting that the brain mechanisms underlying therapeutic effects and secondary effects represent distinct neurobiological processes.
Whole-Brain, Non-invasive Imaging in Awake Animals
The authors highlight the benefits of the fUSI approach over traditional preclinical imaging methods, such as fMRI. Unlike fMRI, which typically requires animal immobilization and sedation or anesthesia potentially confounding brain function studies, fUSI enables imaging in fully awake subjects.
The authors also took advantage of fUSI’s ability to deliver high sensitivity in mice without craniotomy or skull thinning, simplifying experimental procedures.
Another benefit of fUSI is deep-brain imaging capabilities. In this study, the standard coronal imaging plane was set to Bregma –1.8 mm, allowing simultaneous imaging of key regions including the somatosensory cortex, retrosplenial area, dorsal hippocampus and thalamus. As the authors note, “remarkably, this single slice […] yielded already highly significant and reproducible outcomes”.
Advancing the Search into Safer Analgesics
The findings confirm that fUSI-acquired functional connectivity readings serve as a robust, dynamic, and pharmacologically relevant ‘fingerprint’ of opioid drugs action in the brain.
The authors emphasize that this approach “has the potential to contribute to bridging the explanatory gap between cellular/microscale neuronal mechanisms and large-scale neural dynamics that underlie behavior”.
Bruno Osmanski, Ph.D., Co-founder & Chief Scientific Officer at Iconeus, comments: “This exciting and very thorough piece of work adds to the growing number of studies highlighting the role that fUS can play in the development of new pharmaceuticals”.
Specifically, they show that fUS-based imaging of functional connectivity is a useful tool to visualize the effects of drugs on the brain, and so aid the development of new highly-effective analgesics with improved pharmacological profiles. It’s a great illustration of the value of the technique, and we look forward to further studies in this area.
Reference:
J.-C. Mariani, S. Diebolt, L. Beynac, R. Santos, S. Schulz, T. Deffieux, M. Tanter, Z. Lenkei and A. Kliewer, Opioid-specific brain connectivity dynamics distinguish analgesia from secondary effects: Studies in male mice, PNAS, 2026.
DOI: 10.1073/pnas.2505464123