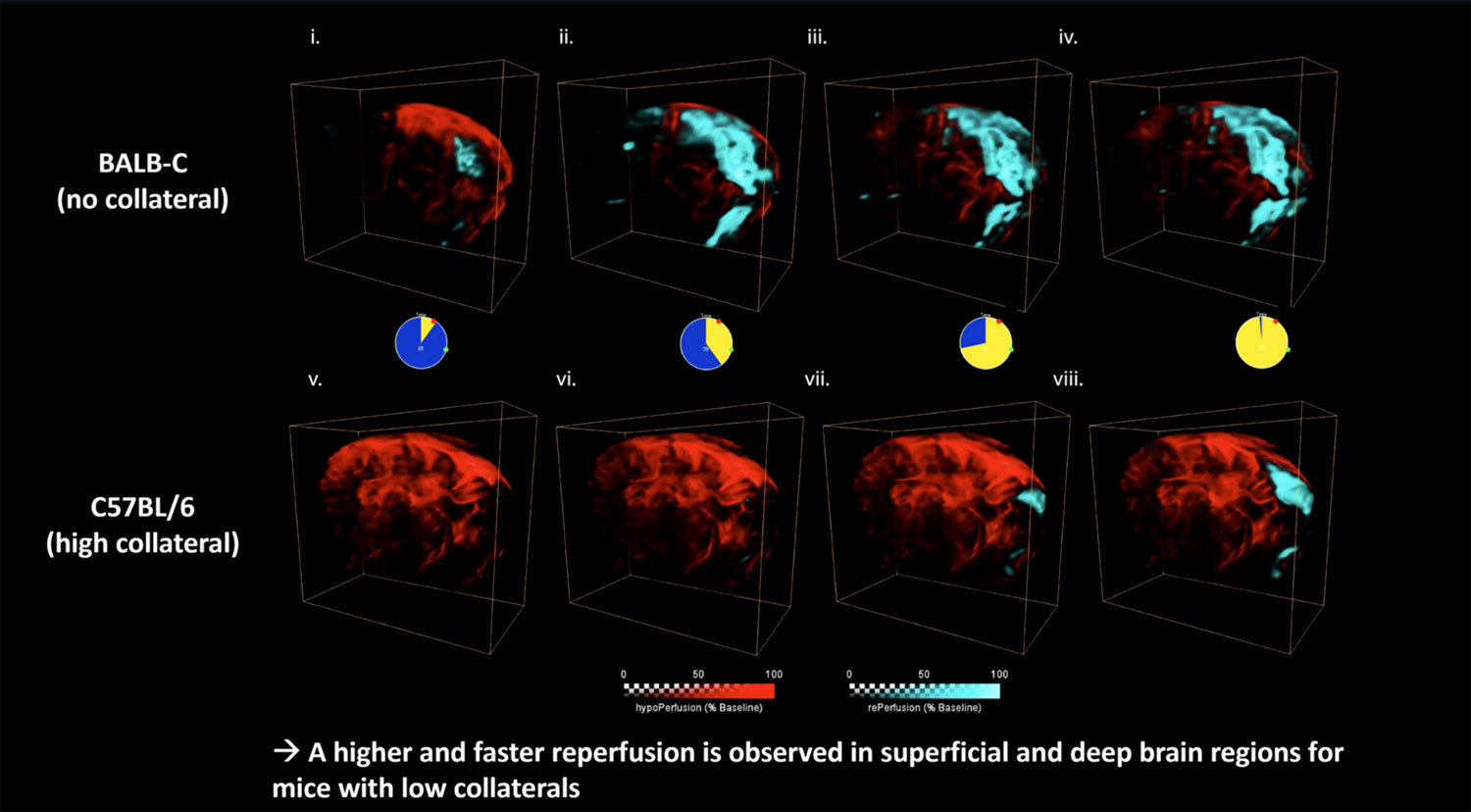
Our existing ultrasound localization microscopy (ULM) technique is a powerful way of generating static images of the brain vasculature at the microscale, using the enhanced signal contrast provided by injected microbubbles. Now, a new paper in Nature Methods authored by our academic partners shows that it’s possible to reduce the temporal resolution to just 1 s, allowing monitoring of changes in blood flow as they happen with a resolution of just 6.5 µm.
Over the past few years we’ve been devoting a lot of time to our ultrasound localization microscopy (ULM) imaging method, which uses injected microbubbles to enhance contrast and so allow imaging of micro-sized blood vessels, as well as offering the potential to overcome the challenge posed by thicker skull bones (as in humans). We’ve been making some great progress, and in April this year we showed how it can be used to image the mouse brain in 3D with a resolution of 20 µm.
Improving the temporal resolution of ULM
To acquire a typical static ULM image with 5–10 µm resolution takes about 60 seconds, resulting in a time-averaged image. But what if that acquisition time could be reduced to a second or so? That’s exactly what our colleagues at Physics for Medicine Paris have been working on as part of the ‘functional ULM’ project, and we’re pleased to say this effort has now borne fruit, in the shape of a paper in the journal Nature Methods.
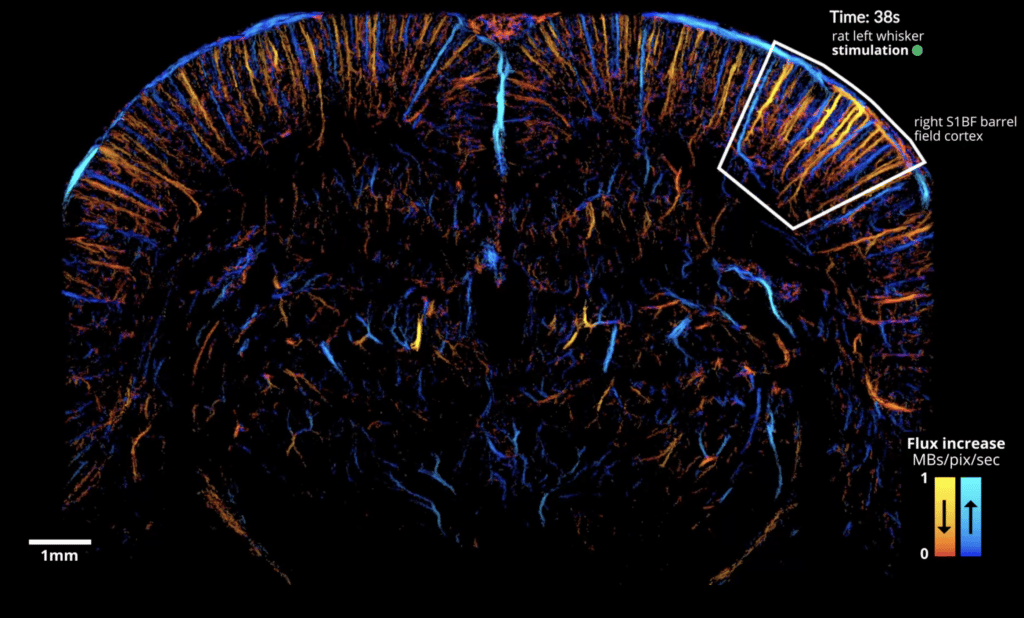
The paper, co-authored by members of the Iconeus team, provides an experimental protocol and data-processing pipeline for dynamic imaging of functional hyperemia (local increases in blood flow related to neuron activity) following whisker or visual stimulation in the rat. Instead of using the whole acquisition dataset to generate static ULM images, the paper reports how it’s possible to use a temporal ‘sliding window’ to construct dynamic maps instead, giving a temporal resolution of just 1 s.
High-resolution functional brain mapping
Importantly, the method is able to image the precapillary arterioles, which feature smooth muscle cells that have been shown to be the main controllers of cerebral blood flow. The results obtained also complement these earlier findings by showing that the relative increase in microbubble flow is actually greater in first-order capillaries and below. It also enables depth-dependent variation in blood speed to be understood, and quantifies the large increases of microbubble flux, blood speed and vessel diameter in venules during activation.
Using fULM, we can also follow large numbers of individual microbubbles, which provides a way of disentangling the spatial extent of downstream or upstream vessels, and allows us to obtain robust estimates for metrics such as microbubble flow speed and flux.
Prospects for fULM
What of the future? Ludovic Lecointre, Pharm.D., CEO and co-founder of Iconeus, provides his perspective:
“This publication is the culmination of a major effort from our partners at Physics for Medicine Paris and the Iconeus team, and a great demonstration of the power of fULM. Being able to visualize changing blood flows at the microscale provides a tool for investigating functional connectivity, layer-specific cortical activity and alterations in neurovascular coupling across the brain – it’s a really exciting development”.
“Applying fULM in mice should be straightforward, and paves the way to using genetically modified models, molecular tools and even spatial RNA-sequencing techniques… the list goes on! But now that this landmark paper has been published, our initial focus will be on implementing the fULM method in our product offering. There are a number of challenges to address before that happens, but we’re confident that they can be overcome, and that it won’t be long before researchers are able to use this powerful method to uncover new insights into the brain using Iconeus One”.
Reference:
N. Renaudin, C. Demené, A. Dizeux, N. Ialy-Radio, S. Pezet and M. Tanter, Functional ultrasound localization microscopy reveals brain-wide neurovascular activity on a microscopic scale, Nature Methods, 2022, 19: 1004–1012, https://doi.org/10.1038/s41592-022-01549-5.